 
These notes cover the basic structure and subatomic particles of an atom, and help students understand the information that can be found on each element’s square of the Periodic Table. I designed these Atoms Cornell Doodle Notes to be an approachable and fun introduction to atoms, subatomic particles, basic atomic structure, and reading the Periodic Table. #4: Atoms Cornell Doodle NotesĪtoms make up everything! And most students do not have a grasp of what an atom is. Also, you can set up a conductivity station with a conductivity apparatus (wires with tiny light bulb attached) or an electric conductivity meter if you don’t have enough of these for each group. 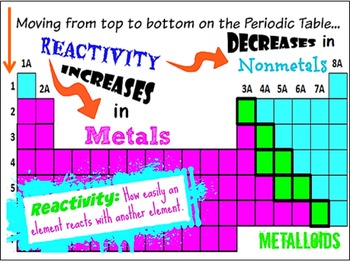
You will also need: a hydrochloric acid solution, a copper (II) chloride solution, a nail, and a Chem plate or small test tubes and a test tube rack for each group. An editable file is included in the download so that you can change the elements if necessary. I wrote the answer key to include aluminum, carbon, copper, magnesium, silicon, sulfur, antimony, calcium, and zinc, but you can use any combination of these elements for the lab. You will need to gather element samples and some basic materials for each group of three students. This is a great inquiry lab that satisfies NGSS standards! Students will be making observations of the physical and chemical properties of pure element samples, grouping them into categories, and classifying them as metals, non-metals, or metalloids. I like to use this Metals, Nonmetals, and Metalloids observation lab early-on in my students’ exploration of the elements and the Periodic Table because students do not need to know about atoms– they only need to know how to make observations and a general sense of the Periodic Table. The student sheet is included as a digital version, too! #3: Metals, Non-metals, and Metalloids Lab The students enjoy looking at the pictures and seeing the elements in a “fresher” way! You could also have your students simply go to the website where the table is located and use this activity more like a webquest. I print a colored class-set of the table front/back (there is more information on the back of the squares), and a worksheet for each student. 
These include the number of valence electrons, atomic radius, number of electron shells, reactivity, boiling and melting points, electron affinity and electronegativity, and more! My most favorite activity to jump-start a unit on Atoms and the Periodic Table is this Periodic People activity. The Periodic Table is an amazing tool because it has SO MANY patterns embedded in it. I love this unit because the growth in my students’ understanding is so significant and they undergo a transformation before my eyes! With the exception of the occasional chemistry-obsessed student, most of my students start out this unit thinking that the Periodic Table is some sort of super complex graphic for brainiacs and mad scientists, or they simply think it’s an absolute bore.īut after a few weeks of study, they can navigate the families of elements on the table, interpret atomic structure based on an element’s position on the table, and glean information about how different atoms will interact with one another! Experiencing my students fall in love with the Periodic Table is amazing every year! In this post, I’m sharing some of my favorite activities to teach my middle school students about the Periodic Table. The unit that I most love to teach is Atoms and the Periodic Table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
